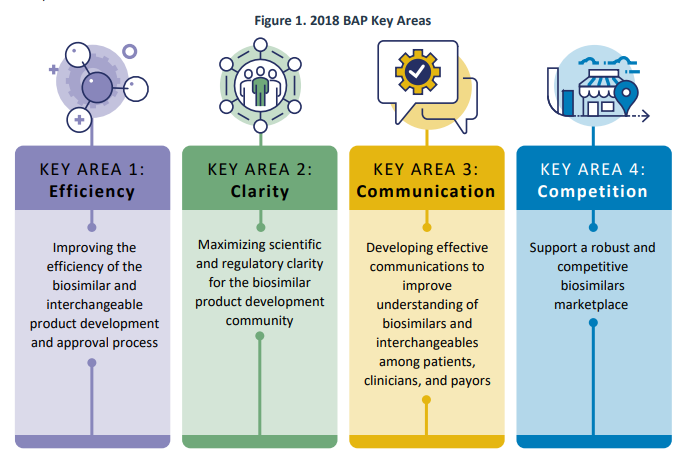
In July 2018, FDA launched the Biosimilars Motion Plan (BAP), which outlined FDA’s method for increasing entry to biosimilars for the American public. The plan targeted on 4 key areas:
A current FDA report evaluations a few of their accomplishments since then. Most of those efforts revolve round steering paperwork, extra workers, schooling merchandise and web sites, public hearings and rules (i.e., proposed/remaining guidelines). FDA additionally added new information assets together with publishing a modernized model of the Purple Ebook in February 2020. FDA additionally collaborated with different businesses equivalent to FTC, and produced a joint assertion and held a workshop in March 2020, entitled: “Public Workshop: FDA/FTC Workshop on a Aggressive Market for Biosimilars.” Another key actions are listed beneath.

The total report is right here.